 The scientists of University of Wisconsin-Madison are in the news again today. Just days after hitting headlines with their dating of a rock that comes from the birth of our planet, the university’s science department is at it again; this time finding a cheaper and more efficient way to produce prototype fuel cells for hydrogen-powered vehicles.
The scientists of University of Wisconsin-Madison are in the news again today. Just days after hitting headlines with their dating of a rock that comes from the birth of our planet, the university’s science department is at it again; this time finding a cheaper and more efficient way to produce prototype fuel cells for hydrogen-powered vehicles.
Hydrogen-powered vehicles are expensive to run; or at least they have been until now. The problem is that the fuel cells used to produce their energy are not cheap to make. But at Madison-Wisconsin, scientists have found a way to harness the hydrogen from the sun to power these as yet rare vehicles; an altogether different form of solar power.
Professor Kyoung-Kim Choi says that the main problem with popularizing or mass producing these vehicles had been that the cost of materials to create their power cells has been prohibitive, while the conversion of solar energy to fuel has not been optimized. Thus the math simply does not work out. But if the materials could be made in a more cost-effective manner and the solar-to-fuel energy output ratio perfected, there would be less of a bar to these alternative energy vehicles being competitive with traditional fossil-fuel transportation.
Choi and another researcher, Tae Woo Kim, may have found a way to do just that. They proved that it was possible to extract hydrogen from water molecules using solar energy, with affordable materials consisting of nickel- and iron-oxide photoelectrodes. This is essentially the same easily-produced technology used to electroplate jewelry. The findings, published in Science, show that record efficiency can be achieved using these relatively cheap methods. Choi and Kim have exceeded all previous models by achieving a 1.7 percent solar-to-hydrogen conversion efficiency.
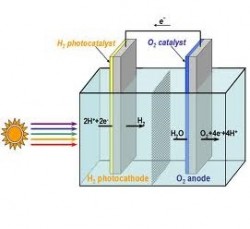 And they did it, in Choi’s words, without any really fancy equipment. They did not need to induce high pressures or temperatures to split the water molecules. Their semiconductor was porous to the nano-level. When made up of such tiny particles, a conductor would have a very high surface area, upwards of 32 square meters for every gram of material, contributing to the efficiency of their results. Their prototype hydrogen fuel cell is cheap, efficient and easily replicable.
And they did it, in Choi’s words, without any really fancy equipment. They did not need to induce high pressures or temperatures to split the water molecules. Their semiconductor was porous to the nano-level. When made up of such tiny particles, a conductor would have a very high surface area, upwards of 32 square meters for every gram of material, contributing to the efficiency of their results. Their prototype hydrogen fuel cell is cheap, efficient and easily replicable.
The semiconductor used in this experiment has as yet received little acclaim from fellow hydrogen-power researchers. Choi acknowledges that their research is only the beginning of a wider study, as the interface between the catalyst of the reaction and the semiconductor that gives it its ‘habitat’ has to be optimized no matter how refined or well-engineered the catalyst and the superconductor happen to be.
Hydrogen fuel cells, therefore, still need a lot of work before enough hydrogen can be extracted simply and cost-effectively enough to make this sort of power system workable in our daily lives. Choi and Kim are not the first persons to use solar energy to split hydrogen atoms, but they are the first to make a solid go of it with such inexpensive materials. Their work proves that record-breaking efficiency does not have to have a huge price tag, and thus moves us all just a bit closer to having affordable hydrogen fuel cells in our lifetimes.
Just proving that such a thing is possible is a leap forward in the research; to have it be so easily repeated and improved upon means that great things are still to come. The work done at the University of Wisconsin-Madison is a promising entry into the field. Perhaps we can soon look forward to a day where cheap and efficient hydrogen fuel cells are available to replace fossil fuels, based on this promising prototype.
By Kat Turner
Sources
University of Wisconsin-Madison News
Discover more from Guardian Liberty Voice
Subscribe to get the latest posts sent to your email.


