Low sex drive and the relationship stress that can come with it could be something of the past if the makers of Flibanserin manage to get their product on the market. The pill, Flibanserin is touted as being the first of its kind to boost women’s sex drive and relieve at least one common argument among couples. However, the magic attitude that researchers have been on a decade-long hunt for may still be a long way away from consumers.
Sprout Pharmaceuticals Inc. submitted their drug for approval but were received with rejection by U.S. regulators. The unanimous decision was agreed upon by all 11 counsel members as Flibanserin lacked evidence of having any more than a placebo affect. However, the scientists behind the drug support its effectiveness with quite a bit of research. Recent statistics have shown that well over 40 percent of women have experienced sexual dysfunction at some point in their relationships. Equally shocking for some people, over 10 percent of women have low sexual desire that is further accompanied by stress within their relationships.
The importance of sexual compatibility would be quite naturally understood by most people and it is because of that, the makers of Flibanserin are so interested in getting it on the markets. (Money might also have something to do with it.) The fight to relieve sexual dysfunction in women is now 15 years behind that of men. Pfizer, the makers of Viagra, have managed to make close to a 4.5 billion dollar market from the male equivalent to Flibanserin and yet even they gave up in 2004 on creating a pill for women.
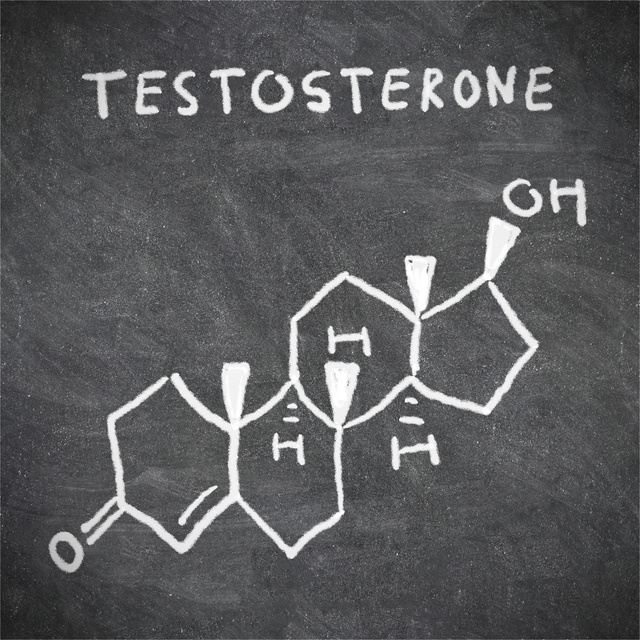
The Mayo Clinic has reported that an increase in testosterone levels in women does seem to have a positive effect on sexual desire in women. The hormone naturally found in females, acts as both a libido developer but is also found to be the trigger hormone for the nipples and clitoris to be sensitive to sexual pleasure. Naturally, this would be a logical place for drug makers to start. Flibanserin follow this research and is what the Food and Drug Administration (FDA) would consider to be a testosterone treatment for women. Currently, the FDA has not approved Flibanserin or any other testosterone treatments for women to boost women’s sex drive.
The great challenge in addressing sexual dysfunction in women is that commonly the root of it is from multiple factors. Currently medical professionals dealing with female patients struggling with the disorder find is that usually depression, unsatisfying relationships and body image tend to have more to do with the issues at hand. Not only is it harder to treat emotional issues with medication, the side effects of many anti-depressants includes a decrease in sex drive. This can be somewhat counter productive when a lot of the emotional stress is coming from lack of satisfaction from a relationship which can then stem from a sexual disconnect.
 The side effects of Flibanserin can also be a little unsexy as well. Dizziness, sleepiness, nausea, and headache were all included as being reportedly experienced by women involved in the drug trials although less than 10 percent of the women experiencing them discontinued the drugs for that reason. The concerns of some medical professionals however is focused on the long-term effects of increasing a naturally found hormone, even if it is within what would be considered safe levels.
The side effects of Flibanserin can also be a little unsexy as well. Dizziness, sleepiness, nausea, and headache were all included as being reportedly experienced by women involved in the drug trials although less than 10 percent of the women experiencing them discontinued the drugs for that reason. The concerns of some medical professionals however is focused on the long-term effects of increasing a naturally found hormone, even if it is within what would be considered safe levels.
As of yet Flibanserin has not been approved by the FDA but an appeal has been made to see whether there is a future in the fight to boos women’s sex drive. Both men and women everywhere will be cheering on scientists as the hunt continues.
By Romana Outerbridge
Discover more from Guardian Liberty Voice
Subscribe to get the latest posts sent to your email.




2 Responses
Flibanserin is non-hormonal. It acts in the brain, not with testosterone. You’re drawing a few conclusions based on thinking flibanserin is hormonal.
Or perhaps we should be cheering A&W for their a propos advertisement at the top of the page…?
I think long term exposure to such drugs could have dangerous consequences for women’s health. I’m glad you mention the concern of medical professionals in your article.