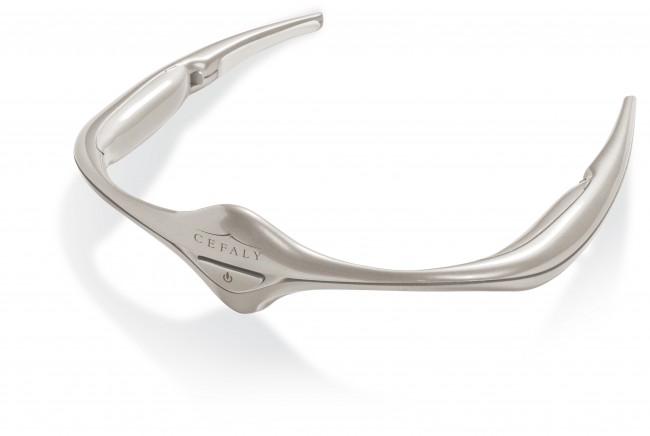
 The Food and Drug Administration has approved a new device that has the ability to help chronic migraine sufferers. The device, called Cefaly, is worn on the head around the eyes and may help make migraines become a thing of the past.
The Food and Drug Administration has approved a new device that has the ability to help chronic migraine sufferers. The device, called Cefaly, is worn on the head around the eyes and may help make migraines become a thing of the past.
The battery-powered gadget is shaped like a tiara and sends out electrodes which can relieve head pain without the need for migraine medications. Much like a pair of sunglasses, Cefaly sits on the ears with a self adhesive band electrode headband. An electric current is then applied to the skin and underlying tissues.
The device, which is currently sold in Canada, will be available only by prescription. The FDA advises that Cefaly should be used only for migraine prevention for up to 20 minutes a day. Users may feel a slight tingling or massaging sensation as the device goes to work at reducing migraine pain and preventing it in the future.
Cefaly was developed by STX-Med, a bioelectronics company whose mission is to provide research and develop innovations for human use, according to the company website. The aim of the company is to focus on health and well-being and is composed of a team of physicians and engineers based in Belgium. STX-Med’s latest mission is to make the migraine become a thing of the past with Cefaly.
There are nearly 37 million migraine sufferers in the United States alone. A migraine typically involves pain on either side of the head, sensitivity to light, nausea and occasional vomiting. It is estimated that migraines cost the U.S. $20 billion a year in doctor visits, medical expenses, medication, and less directly, when employees miss work.
A recent study has suggested that a constant migraine can actually change the brain. Dr. Messoud Ashina, a neurologist at the University of Copenhagen in Denmark and author of the study said that migraines may “permanently alter brain structure,” in more ways than one. Using magnetic resonance imaging, or MRI, the study was able to detect changes in the brain after a migraine that X-rays may not detect. The study found that those who suffer from migraines have an increased chance of white matter brain lesions compared with those who do not suffer from migraines.
Researches also found other white matter brain abnormalities, and cited that more research is needed to understand why this happens in the brain with migraines. Dr. Eishi Asano, professor of neurology at Wayne State University in Detroit, claims that new solutions are needed for migraine prevention. “Further studies of neurostimulation using innovative study designs are warranted to explore the optimum way to create an acceptable evidence base for widespread use of this potentially valuable treatment,” he said.
In a trial published in Neurology, 38 percent of those who used Cefaly found at least a 50 percent reduction in migraine occurrence. While the device has not been proven to reduce the severity of a migraine, it did reduce the number of migraines in the test study by two per month. Unlike prescribed drugs, Cefaly does not have any side effects like most migraine medications. The FDA approval of Cefaly could make the migraine become a thing of the past. The device currently sells for around $350.
By Nathan Rohenkohl
Sources:
Discover more from Guardian Liberty Voice
Subscribe to get the latest posts sent to your email.



3 Responses
I agree with mythfyt. As someone who actually works in clinical trials for a living, my professional opinion is that this article is horribly written.
I am very VERY interested in doing a trial of this, but their website will not sell to me as I am in America. Does anyone know when, or HOW I can order this??? If push comes to shove I can find someone in Canada to cross ship it to me, but that’s another headache I just dont need.
I am tired of missing work (so is my boss) as well as needing to take enough medication that would kill a fair sized elephant. I have lived with this for ten years (since I was 22 years old) and I just want to be a somewhat normal person again.
If anyone has any information at all about obtaining it in the USA please email me or respond to this post! I emailed the company since I couldn’t get through their 1-800 number due to being in the USA.
email is [email protected]
38% of users had a reduction of 50% … reduced number of headaches by 2 per month…does make the “migraine a thing of the past.” ONE possibly helpful tool for some people, yes. How about some accuracy in reporting, please?