The war on drugs in the U.S. had begun in 1971 and while most drugs are illegal in the United States, the nation is currently facing a heroin epidemic. To help emergency room workers battle one of the country’s worst enemies, an antidote for heroin is under development to take on a nasal spray form.
A doctor from Lexington, Kentucky has signed a partnership agreement with the popular pharmaceutical company, Reckitt Benckiser Pharmaceuticals Inc.. The University of Kentucky professor plans to get life-saving heroin antidote in the hands of the paramedics and emergency room workers.
Dr. Daniel Wermeling is also the founder of AntiOp Inc., which is a special pharmaceutical company developing naloxone nasal spray. The product is entering its final clinical trial in June.
Signing an agreement with Reckitt Benckiser Pharmaceuticals Inc. is meant to accelerate both the marketing and the production of naloxone, which is designed to treat opioid and heroin overdoses. According to Wermeling, the partnership will combine the strengths of two companies.
The product cannot come soon enough for American hospitals. Roughly 16,500 Americans die each year from abuse of different prescription opioids,this includes some prescription painkillers such as methadone, oxymorphone, hydrocodone and oxycodone. The drugs are chemically similar to heroin, and abuse of the drugs has been has fueled by the recent heroin addiction crisis.
According to the National Institute on Drug Addiction, there are between 300,000 to 500,000 heroin addicts in the United States. This is 75 percent more than the population estimates from just five years ago.
In the past 10 years, heroin and related opioid pain pills have killed more over 125,000 American citizens, according to Centers for Disease Control and Prevention (CDC) data. These numbers are only expected to increase.
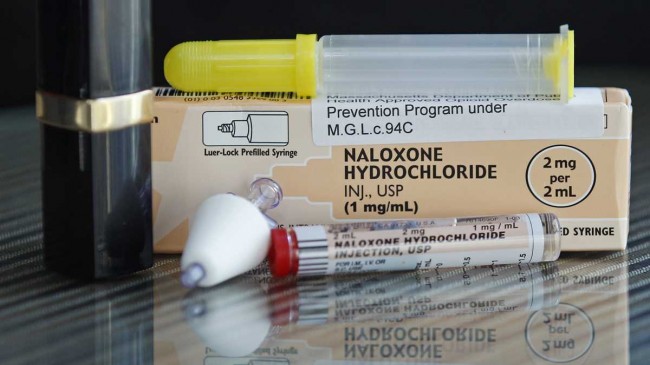
According to Wermeling, naloxone injections are common in emergency rooms, ambulances and post-surgery recovery rooms across the country. The currently used drug is injected using a variety of methods including muscle tissue injection, intravenous, and under the skin method. This has not stopped many emergency workers from getting creative, they have been using atomizers to convert the injectable form of naloxone into a nasal spray. This creativity had given Wermeling an idea to develop the heroin antidote nasal spray.
Wermeling’s product improves on this idea, and allows rescue treatment when the patient is unconscious. The new nasal spray will be administered using a single-use and ready-to-use delivery device. Following the insertion of the device into the nose of the patient, the device delivers a consistent dose of naloxone. The drug can even enter the bloodstream of the patient if the patient is not breathing through absorption across the nasal membranes. Nasal spray use eliminates the need for needles, is safer and disposable.
Wermeling heavily relies on customer feedback. In an interview, he had stated that he is open to any comments regarding his product. Intranasal naloxone is created to serve the users and stakeholders, and it is important to Wermeling developing the standard of care in opioid overdose cases.
While the heroin antidote nasal spray is still under development, Wermeling is excited for the drug’s eventual release. The final clinical study that will start in June, will focus on measuring blood levels of naloxone in users of the intranasal product compared to products already on the market.
By Ivelina Kunina
Sources:
USA Today
Detroit Free Press
New York Daily News
Discover more from Guardian Liberty Voice
Subscribe to get the latest posts sent to your email.


