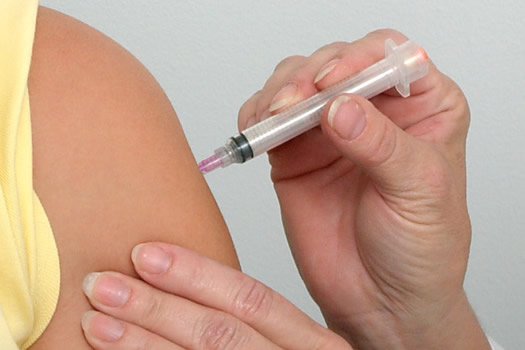
For the 2013 to 2014 flu season, CDC recommends two types of new novel design influenza vaccines, a trivalent formulation (protects from three virus strains), and a newer quadrivalent formulation (protects from four virus strains).
Both vaccines offer immunity against three most common strains of influenza:
• Type A – California/7/2009 (H1N1) like virus
• Type A – Victoria/361/2011 (H3N2) like virus
• Type B – Massachusetts/2/2012 like virus
However, the quadrivalent vaccine offers immunity against a fourth strain of newly identified influenza virus, the Type B/Brisbane/60/2008-like virus. This formulation is manufactured by Sanofi Pasteur, with the brand-name Fluzone Quadrivalent.
Unlike previous years, the vaccine manufacturers came up with a novelty in the design of vaccine formulations and their delivery techniques. They formulated vaccines to meet the unmet needs in the clinical ecosystem using innovative methods.
Egg less vaccine for people allergic to egg albumin
As October started, there is an increased buzz about flu vaccination. This is not surprising as October marks the start of the flu season in the U.S. as per Centers for Disease Control and Prevention (CDC). At this time of the year, many hospitals and community health authorities will begin their vaccination campaigns.
Each year, U.S. Food and Drugs Administration approve influenza vaccines for a specific flu season, and along with CDC, it updates the recommendations on the type of influenza virus strain that should be comprised in the formulation of these vaccines.
Flublok is a new unique formulation specifically designed to deliver the vaccine to people who are allergic to eggs. Protein Sciences, the company which manufactures Flublok, announced that their egg less vaccine only contained the hemagglutinin protein isolated from the virus, instead of using the whole virus cultured in the chicken embryo, as observed in other commercially available vaccines. They incorporated the gene coding the hemagglutinin protein into a virus which can infect insect cells, which are cultivated to produce the antigen protein for developing Flublok.
Flublok was intended to be used only in healthy adults around the age group of 18 – 49 years old.
High-dose vaccine for elderly
The standard dose of the influenza vaccine is not completely effective in elderly people because of their weaker immune system. However, Sanofi Pasteur provided the solution to this issue by developing a high-dose vaccine.
A clinical study conducted in 30,000 elderly people, demonstrated that the high-dose vaccine offers 24.2% higher immunity compared to the standard dose vaccine.
This high-dose formulation is marketed under the brand-name Fluzone High Dose and is intended to be used only in people over age of 65 years.
Painless Intradermal vaccine
An Intradermal formulation of the trivalent vaccine was developed to deliver the vaccine through skin instead of an intramuscular injection. This formulation uses ultra-thin, micro needles for painless vaccine administration. FDA approved this formulation with a brand-name Fluzone Intradermal for use in people, in the age group 18 – 64 years.
Generally, FDA approved a total of seven manufacturers for developing influenza vaccines for the 2013 to 2014 flu seasons. Now it is the duty of the healthcare professionals to discuss a suitable formulation with their patients, and assist them in undergoing vaccination.
Written by: Janet Grace Ortigas
Sources: FDA CDC Healthline
Discover more from Guardian Liberty Voice
Subscribe to get the latest posts sent to your email.



4 Responses
Are the available vaccines effective in both Canada and the US. Are there any differences
where is the vaccine manufactured?
I BET THERE IS STILL A CHANCE OF GETTING GUILLIAN BARRE AFTER A FLU SHOT ..
PROVE THERE IS NOT????
Thanks to Janet for the Info.
I do not know and will find out whether in Indonesia already exists influenza vaccination.
thank you all… Lets ride your bicyle everyday….