
Investors are singing the praises for Orexigen Therapeutics, Inc. This morning as the company announced a resounding success concerning the prospects of an obesity drug currently under development.
Contrave is intended to be a treatment for poorly managed obesity. The drug is a combination of two medications. Buproprion, which is as fun to say as it is to spell, increases dopamine secretion in order to stimulate the neurons in the brain that regulate appetite and metabolism. Naltrexone, an opioid antagonist normally used to treat addiction, plugs the negative feedback loop that is built into these neurons so that they never realize that their job is finished and keep working. The net result is that users are expected to feel less hungry and to burn more fat through activity than they normally would.
Orexigen Therapeutics, Inc. first submitted a New Drug Application (NDA) to the Food and Drug Administration (FDA) in 2011. Federal regulators at that time expressed concerns over the possibility of Contrave producing adverse cardiovascular effects, that would essentially cancel out the intended health benefits of treating obesity. The FDA outlined a set of criteria to establish the safety of the drug and advised the pharmaceutical company that, should the standards be met, the drug would likely be approved.
 The eagerly anticipated milestone arrived this morning, as Orexigen announced that “an unprecedented amount of cardiovascular outcomes data” has confirmed that Contrave presents no discernible cardiac risk. The drugmaker, heartened by its findings, intends to submit its NDA once again, whereupon the FDA is expected to honor its promise of approval.
The eagerly anticipated milestone arrived this morning, as Orexigen announced that “an unprecedented amount of cardiovascular outcomes data” has confirmed that Contrave presents no discernible cardiac risk. The drugmaker, heartened by its findings, intends to submit its NDA once again, whereupon the FDA is expected to honor its promise of approval.
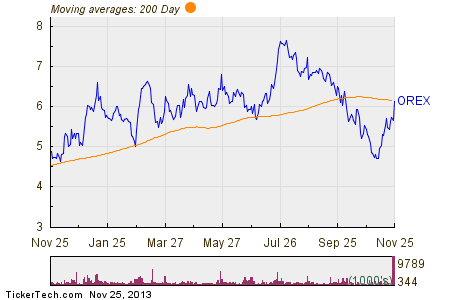
Although getting a green light from the FDA is a lengthy process, investors have examined the prognosis and are clamoring to capitalize on Orexigen’s report on the prospects of its new weight-loss drug. Anticipation has been slowly building as the company’s shares climbed 8 percent over the fiscal year. Today, upon publication of the report immediately before market opening, shares jumped 13 percent to a price of $6.41. That number – which represents a 44 percent climb from its lowest point in the previous year – is expected to continue to rise, which has placed the stock on various watch lists and investor alerts.
The resounding interest in Orexigen is all the more interesting because Contrave would become its first official product for public consumption. Few opening salvos could be better aimed, however, as obesity is the number one health issue facing first world nations. The Centers for Disease Control and Prevention reports that one-third of all adult Americans are morbidly overweight. Obesity causes costly and debilitating health issues, such as vastly increased probabilities of heart attack, stroke, and type II diabetes mellitus. The price tag of obesity-related disease and disorders tops $147 billion annually.
Orexigen Therapeutics, Inc. hopes that Contrave will receive FDA approval as early as July of 2014. There is no word as of yet what the final cost may be to consumers, but investors are pegging their hopes on the prospects of the weight-loss drug. The front-runner in the fight against obesity is expected to see wide use; thus a lot of wallets are going to get fat as an obese public tries to get skinny.
By Daniel Annear
Sources:
SFGate
CDC
Orexigen Therapeutics, Inc.
4-Traders
Discover more from Guardian Liberty Voice
Subscribe to get the latest posts sent to your email.



One Response
best program to Lose Fat Naturally