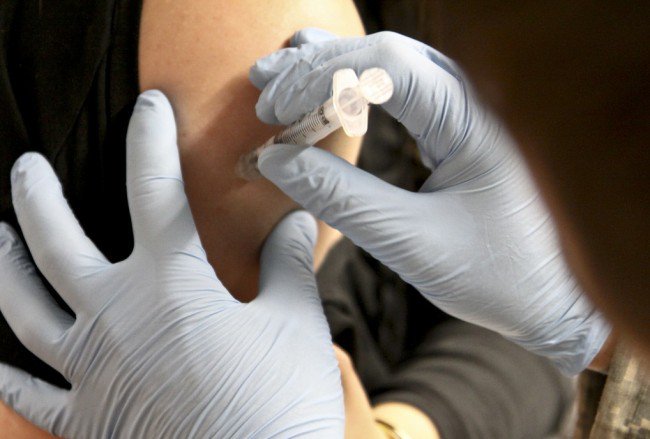
After promising results from animal studies, Ebola vaccine trials with human subjects have begun. Human testing of new vaccines normally lasts for years, but the current crisis in West Africa is so great that the testing of new experimental vaccines is being fast-tracked, so much so that the drug could potentially be in use by the end of the year.
Sixty healthy volunteers are being injected with the vaccine at a safety trial being conducted at the University of Oxford in the UK. Another trial, using a different formulation of the vaccine and using 20 volunteers, is taking place at a National Institutes of Health (NIH) facility in Bethesda, Md. A third trial will begin in Africa next month.
The studies will attempt to determine whether the vaccine produces a good immune response with few side-effects. Blood tests will be taken from the volunteers within two to four weeks to determine the extent of their antibody response against the Ebola virus. NIH Institute of Allergy and Infectious Diseases director Anthony Fauci testitifed before Congress yesterday and said that so far there have been no “red flags” in reactions to the drug. Trials conducted on animals showed that the vaccine provided protection from the Ebola virus without significant adverse side effects.
The vaccine has been co-developed by the NIH and the British drug manufacturer GlaxoSmithKline (GSK). It is based on a cold virus from chimpanzees, chimp adenovirus type 3. The virus is being used to carry genetic material from two different Ebola strains, Zaire and Sudan. It is the Zaire strain causing the current epidemic in West Africa. The vaccine does not contain infectious virus material, so will not cause a person taking the medication to become infected.
With nearly 5,000 people infected with Ebola in Sierra Leone, Guinea, Liberia, Senegal and Nigeria and more than 2,400 deaths since the outbreak began earlier this year, development of a vaccine against the virus is considered critical. Dr. Edward Wright, one of the experts involved in the UK trials, said the disease is difficult to attack without a vaccine because of the rate at which it multiplies in the body. He said that victims of the disease are unable to fight off the rapidly replicating virus, as it moves so quickly that the body is unable to generate enough antibodies to block it before it can take hold. The vaccine is hoped to stimulate production of these antibodies prior to infection setting in.
The trials began Sept. 2 at the NIH Clinical Center in Bethesda. They begin in the UK today. If the trials are successful the drug will be tested in Mali and Gambia to ensure that differences between European and West African people are taken into account by the studies.
If the human trials are successful the Ebola vaccine could be used to immunize health workers by the end of the year, at which time about 10,000 doses should be available. Professor Adrian Hill, who is director of the Jenner Institute in Oxford that is leading the trial, says that this is a “remarkable example” of how quickly new vaccines can be put into use using international cooperation.
By Beth A. Balen
Sources:
BBC
The Telegraph
The Washington Post
Bidness Etc.
U.S. Army Corps of Engineers Photo
Discover more from Guardian Liberty Voice
Subscribe to get the latest posts sent to your email.



One Response
Super speedy! So glad that the rules are being broken in desperate times- hopefully lives will be saved!